 
The other of atomic numbers in range 51-57 and atomic mass in range 125-60.Īlso, fission of uranium is also possible with high speed protons of energy $6. One of atomic number in range 35-43 and atomic mass in range 80-110, and In any case, the uranium fission products fall into two groups: Note that the products of uranium fission are not necessarily always barium and krypton. Nuclear power is the use of nuclear reactions to produce electricity.Nuclear power can be obtained from nuclear fission, nuclear decay and nuclear fusion reactions. 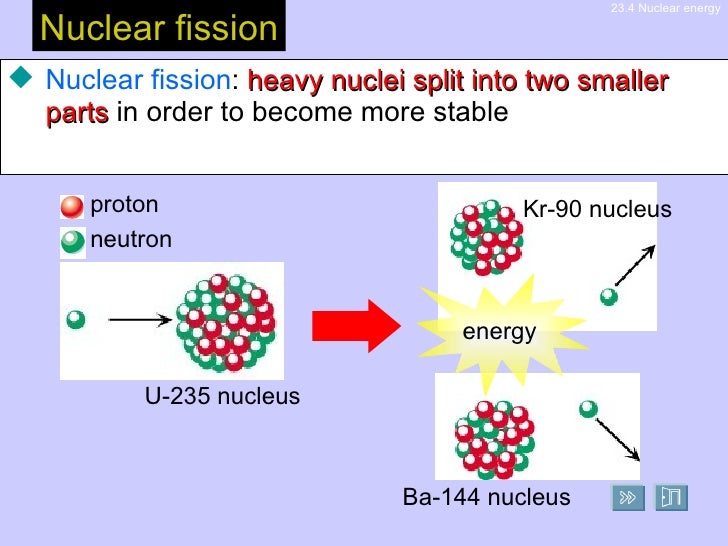
If these reactions are uncontrolled, they result in explosive fission processes, like in atom bombs. The Leibstadt Nuclear Power Plant in Switzerland Growth of worldwide nuclear power generation. Those can hit other nearby uranium-235 atoms and cause those to fission. The number of neutrons produced increases in geometric progression, which in turn split a larger number of uranium atoms, releasing even more enormous amounts of energy in a short time. For example, when a nucleus such as uranium-235 fissions, it emits neutrons. They bombarded a uranium nucleus $^U$ following which the whole process repeats as described above, producing neutrons that are again bombarded with uranium nuclei, producing a perpetual chain reaction. 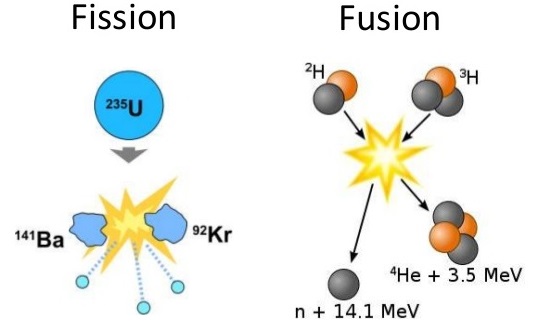
This was essentially how nuclear fission was first discovered by Hahn and Strassman in 1939. Let us look at how this would work with an example of a Uranium atom bombarded by a neutron. Most reactions involve collision of a subatomic particle with an atomic nucleus that results in the formation of resultant fragments and particles. For heavy nuclides with an atomic number higher than 90, most. These materials are used to fuel thermal nuclear reactors because they can sustain a nuclear fission chain reaction. In controlled environments such as reactors, all nuclear fission occurs as a nuclear reaction that is bombardment-driven and involves collision of two subatomic particles. In nuclear engineering, fissile material (nuclide) is material that is capable of undergoing fission reaction after absorbing thermal ( slow or low energy) neutrons. In any case, fission processes are exothermic reactions releasing energy in the form of electromagnetic radiation as well as kinetic energy imparted to the produced fragments. This is made possible through the use of a nuclear reaction. This process can occur naturally by spontaneous splitting of an atom via radiative decay or can be simulated in a lab by achieving appropriate conditions such as bombarding the heavy atom with subatomic particles like neutrons. For example Uranium-235, which is a naturally occurring isotope of Uranium. The phenomenon of splitting up of a heavy atomic nucleus into lighter fragments and nuclei along with the emission of gamma rays or photons releasing large amounts of energy is termed as nuclear fission. As for the nuclear reaction, ensure that it is balanced at all stages. Remember that fission processes usually involve an exudence of a large amount of energy in various forms. In such a case, recall how this would be carried out in case of a uranium atom that is bombarded by a neutron, and the subsequent processes that follow through. Hint: We know that nuclear fission is a type of a nuclear reaction that involves splitting of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
